Connective Tissue Graft: The Biology Behind the Procedure

Table of Contents
A few years ago I decided to learn how to make sashimi at home.
I did what most of us do when we want to learn something seriously. I went online, read the reviews, and bought the right knife. A Yanagiba. The kind with the single-bevel edge that you are supposed to use for fish. I bought a book. I watched videos of Japanese chefs working a piece of tuna like it was a musical instrument. I had the setup. I had the instructions. I had every reason to succeed.
The first cut tore the flesh. So did the second. By the tenth attempt the fish on my board looked nothing like the photographs in the book. The texture was wrong, the surface was ragged, the slices fell apart on the plate. I ended up ordering Japanese food from a restaurant down the street and ate it on the couch.
The knife was not the problem. The book was not the problem. I did not understand the fish. I did not know how the muscle grain ran through that specific cut, how temperature changed its resistance, how to feel tension under the blade and adjust the angle in real time. I had memorized the procedure without understanding the biology underneath it.
I think about this every time someone asks me how to harvest a connective tissue graft.
We buy the right microsurgical kit. We take the courses. We learn the protocol. And then we wonder why the graft does not behave the way the textbook said it would. We blame the technique. We blame the patient. We rarely blame the thing that was missing from the start, which is a clear understanding of what a connective tissue graft actually is, biologically, before our scalpel ever touches it.
What is a connective tissue graft
A connective tissue graft, often abbreviated CTG, is a piece of subepithelial tissue harvested from the palate and transplanted into the recipient site to cover an exposed root surface, thicken keratinized gingiva, or augment soft tissue volume around a tooth or implant. The donor site is most often the lateral palatal region between the canine and the first molar, and the typical graft measures roughly 8 to 15 mm in length and 1.0 to 1.5 mm in thickness.
That is the clinical definition. Underneath it is something most courses skip.
A connective tissue graft is not white tissue. It is organized biology.
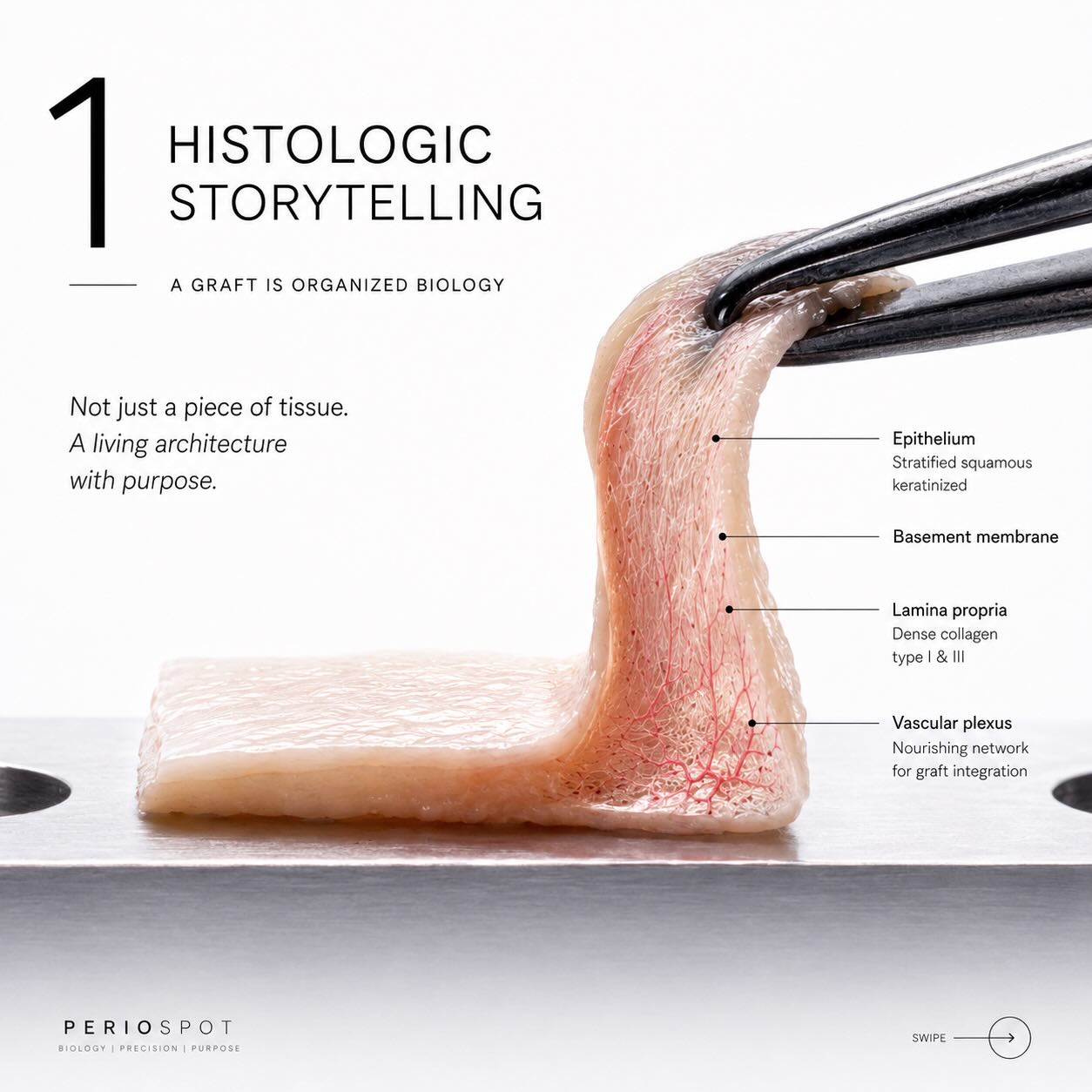
A connective tissue graft is organized biology, not just white tissue. Every millimeter carries biologic information.
This is the central idea of this article. Every clinical decision you make about a CTG, from the harvest technique to the deepithelialization method to the way you handle the graft between donor and recipient, is a decision about biology. The instruments are how we touch the tissue. The biology is what determines whether the tissue heals.
The five biological components of a CTG
A palatal soft tissue graft brings five biologically relevant elements into the conversation. Depending on the harvest and deepithelialization method, the final graft may intentionally exclude the epithelial layer or carry epithelial remnants. Zuhr, Bäumer, and Hürzeler laid out the most useful clinical map of this in their 2014 critical review (Zuhr et al., 2014). Each component has a role in how the graft integrates.

Epithelium, collagen, vascular plexuses, fibroblasts and extracellular matrix, the five components that decide how a graft will heal.
The first component is the epithelium. In a classic subepithelial CTG, the goal is to leave the epithelium at the donor site. In a deepithelialized free gingival graft, however, epithelial remnants can remain on the graft surface. Those remnants are not biologically inert. They can influence how the recipient surface heals over the graft and how visible the result is at the gingival margin.
The second is collagen, organized in bundles that give the graft its mechanical stability. The orientation and density of these bundles vary along the palatal donor zone, which is one of the reasons grafts harvested from different palatal regions behave differently.
The third is the vascular plexus, the network of small vessels that perfused the tissue in its original location. The plexus disappears with harvesting, but its architecture leaves behind channels that the recipient bed will use during revascularization.
The fourth is the fibroblast population. These are the cells that will remodel the graft once it is placed. They synthesize new collagen, reorganize the extracellular matrix, and ultimately determine whether the grafted area looks and behaves like keratinized gingiva months later.
The fifth is the extracellular matrix itself, the molecular scaffold that holds the other four together. It is the part of the graft that most directly determines biological behavior, and it is also the part that is most affected by how you handle the graft during surgery.
This last point matters more than most clinicians realize. The longer a graft sits dehydrated on a tray, the more its extracellular matrix degrades. Mechanical handling of the graft is biological handling.
Subepithelial CTG vs free gingival graft vs deepithelialized CTG
The three most common soft tissue graft types confuse a lot of clinicians, and the search results for these terms are full of patient-facing copy that does not actually explain when to choose one over the other. Here is the clinical comparison.
| Feature | Subepithelial CTG (SCTG) | Free Gingival Graft (FGG) | Deepithelialized CTG (DFGG) |
|---|---|---|---|
| Harvest | Trap-door or single-incision approach, epithelium left at the donor site | Full-thickness with epithelium, taken as one piece | Full-thickness harvest, then epithelium removed extraorally or intraorally |
| Graft composition | Connective tissue only | Epithelium plus connective tissue | Connective tissue, with residual epithelial remnants |
| Donor site healing | Closed by primary intention, low morbidity | Open secondary intention, higher morbidity | Closed by primary intention, low morbidity |
| Esthetic blend at recipient | Excellent, no surface keratin from graft | Visible keratin patch, possible color mismatch | Excellent in most cases, occasional epithelial cysts reported |
| Best indication | Root coverage in the esthetic zone, soft tissue augmentation around implants | Increasing keratinized tissue width in non-esthetic areas | Cases where harvesting a thin SCTG is anatomically difficult |
| My clinical preference | First choice for esthetic root coverage and implant soft tissue | Reserved for non-esthetic keratinized tissue gain | Strong option when the palate is thin and a clean SCTG is hard to harvest |
The deepithelialized CTG is the option that sits in the middle, and it is also the one this article spends the most time on because the biology of deepithelialization is the place clinicians underestimate most often.
If you want a step-by-step procedural breakdown of the four classic harvest approaches, that ground is covered in 4 connective tissue harvesting techniques that may help you in your daily practice. This article is the biology companion to that piece.
Deepithelialization, the silent variable
Deepithelialization sounds like a small step. Lift the graft, scrape or trim the epithelium, place the graft into the recipient site. Three minutes of work between two more dramatic moments of the surgery.

Deepithelialization changes how the graft heals, not how it looks. Different methods leave different amounts of epithelium behind.
It is also one of the most consequential decisions in soft tissue surgery, because the choice of deepithelialization method changes how much epithelium actually leaves the graft, and how much stays behind to influence healing.
The numbers are striking. A 2021 histologic case series by Maia and colleagues examined grafts harvested using the Harris technique and found that only about 56% of the graft surface was free of epithelium after conventional deepithelialization, meaning roughly 44% of the surface still carried epithelial remnants when the graft was placed into the recipient site (Maia et al., 2021).
A 2024 cadaver study by Bara-Gaseni and colleagues compared three deepithelialization methods directly and produced the cleanest data we have on the question. Using an intraoral bone scraper, 97.98% of the graft surface was free of epithelium. With an intraoral diamond bur, 88.24%. With an extraoral scalpel, the workhorse method most of us learned, only 58.84% (Bara-Gaseni et al., 2024).
Three things follow from this data.
First, if you are using a scalpel extraorally and you assume your graft is clean, you are wrong roughly four times out of ten. The epithelium you cannot see clinically is there histologically.
Second, the method matters as much as the operator. A bone scraper is not just a tool with a different handle. It produces a graft with a different biological profile.
Third, residual epithelium does not always cause problems, but when it does, it shows up as small surface irregularities, occasional epithelial cysts, and a graft that takes longer to mature in tone and texture (Maia et al., 2021). The biological signal under the clinical surface.
This is exactly the kind of insight you cannot get from a textbook procedure list. It only appears under the microscope, which is where the rest of this article is going to take you.
Revascularization and integration
The most important thing that happens to a connective tissue graft happens in the first 7 to 14 days, and almost none of it is visible to the naked eye.
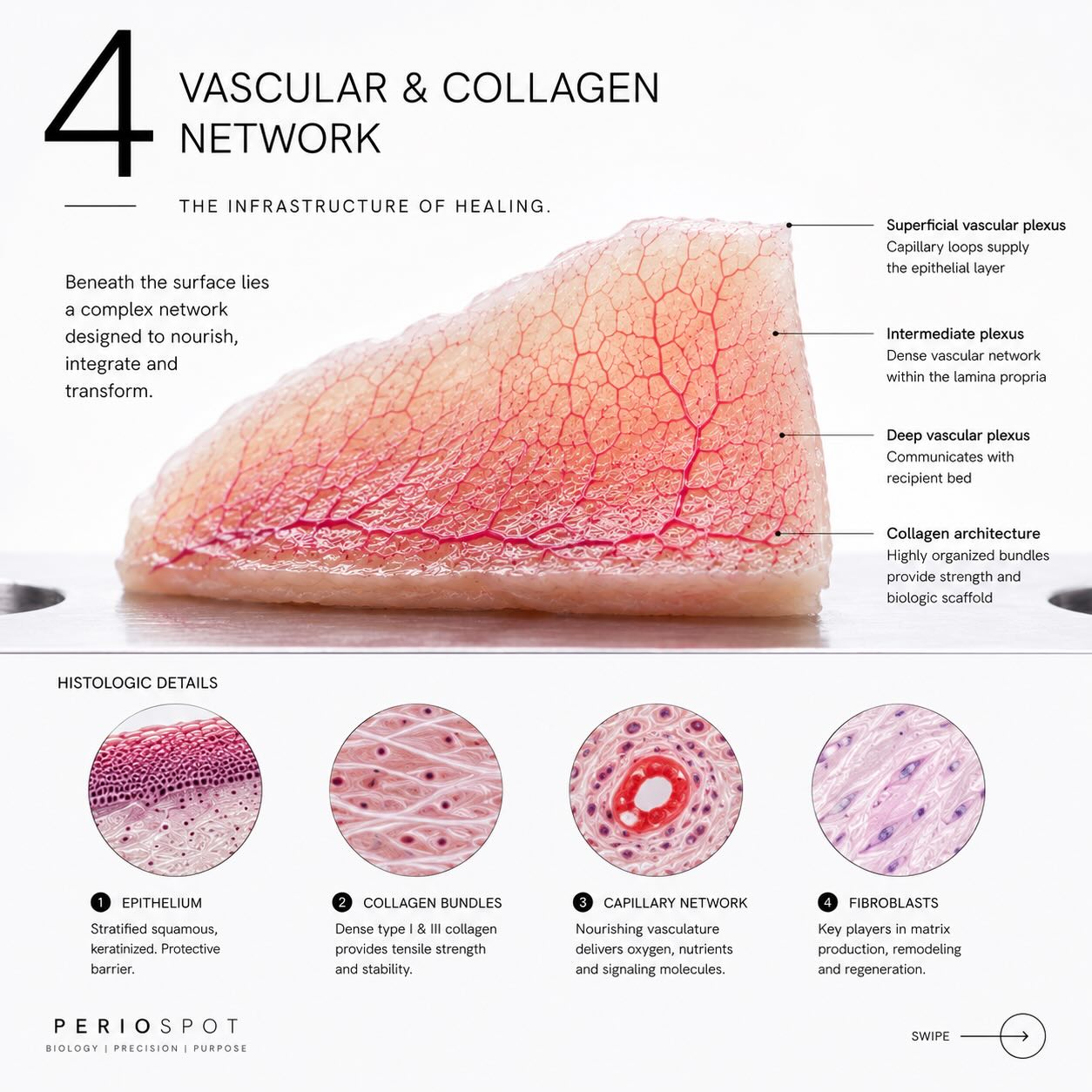
Plasmatic circulation, capillary inosculation, and fibroblast remodeling. The biological sequence that turns harvested tissue into integrated gingiva.
When a graft is placed into a recipient bed, it is, for a few days, biologically isolated. It has no blood supply. The cells in the graft are alive but starving. They survive on plasmatic circulation, a process in which nutrients and oxygen diffuse from the surrounding tissue fluid into the graft through passive transport. This phase typically lasts 48 to 72 hours and is the moment of greatest risk for graft failure. Anything that disrupts intimate contact between the graft and the recipient bed during this window, including hematoma, micromovement, or insufficient flap closure, can lead to graft necrosis.
The second phase is capillary inosculation. Vessels from the recipient bed begin to grow toward the cut ends of the vessels that previously perfused the graft. By day 4 to 7, fresh capillary loops are forming inside the graft. The vascular plexus that disappeared with harvesting is being rebuilt from below, using the channels the original architecture left behind.
By day 7 to 14, the graft has its own blood supply. Fibroblasts begin migrating through the matrix, remodeling collagen, and laying down new tissue that gradually replaces the original graft material. This phase continues for months. The clinical appearance of the gingival margin may look mature at week 4, but the histologic process of integration is still happening at week 12 and beyond (Tavelli et al., 2019).
This is also the biological reason why pressure on the recipient site, smoking, and aggressive brushing during the early healing period are so destructive. They interrupt the very specific sequence of plasmatic nutrition, inosculation, and remodeling that turns a piece of harvested tissue into living, integrated gingiva.
When patients ask me how long a connective tissue graft takes to heal, I give two answers. The clinical answer is six to eight weeks. The biological answer is six months.
Epithelial remnants under the microscope

What looks clean at the chairside often still carries epithelial islands at the histologic level.
The Maia 2021 case series and the Bara-Gaseni 2024 cadaver study both make the same broader point through histology: what we see clinically and what is actually happening at the cellular level are not the same thing.
Under the microscope, a graft that looks perfectly deepithelialized at the chairside often still carries islands of epithelial cells embedded in the connective tissue. These epithelial remnants are usually clinically silent. Occasionally they form small inclusion cysts in the months after grafting, visible as small bumps or surface irregularities. More often they simply influence the rate at which the graft tone matures and blends with the surrounding gingiva.
This is the part of soft tissue surgery that does not fit on a slide in a course. It only becomes visible when histology becomes part of the educational vocabulary. And it is the reason I keep coming back to the same thesis. Procedure is what we do. Biology is what determines the outcome.
Non-autogenous alternatives
Autogenous connective tissue is the gold standard. It is not the only option.
In the last fifteen years a series of xenogeneic and allogeneic soft tissue replacement materials have entered the clinical workflow. The two best documented categories are xenogeneic collagen matrices, such as Geistlich Mucograft and Fibro-Gide, and acellular dermal matrices of human or porcine origin. These materials trade some biological behavior for the elimination of a second surgical site.
The clinical picture from the systematic review evidence is consistent. For root coverage, autogenous CTG still produces slightly better mean root coverage and slightly better keratinized tissue gain. For increasing soft tissue thickness around implants, xenogeneic matrices perform within a clinically acceptable range and significantly reduce patient morbidity (Tavelli et al., 2019).
The biology of a non-autogenous graft is different. Without living fibroblasts inside the matrix, integration depends entirely on cell migration from the recipient bed into the scaffold. The matrix is consumed over months and replaced by host tissue. The end result can be excellent, but the timeline and the failure modes are not the same as for an autogenous CTG.
In my practice the decision is not "autogenous versus non-autogenous." It is "what does this specific patient and this specific defect need?" Esthetic zone root coverage in a thin biotype with a single recession defect, autogenous CTG. Soft tissue thickening around three adjacent implants in a heavy-set posterior maxilla, often a xenogeneic matrix. Most cases live somewhere in between, and the decision is biological as much as clinical.
Healing timeline and patient recovery
Most patients want a calendar more than a histology lecture. Here is the timeline I give them.
Day 0 to 2. The graft is surviving on plasmatic nutrition. Cold packs, soft diet, no rinsing. Most of the swelling and tenderness happens in this window.
Day 3 to 7. Capillary inosculation begins. The graft starts receiving its own blood supply. Patients describe a sense of tightness in the area, which is healing tension, not infection. Sutures usually remain in place.
Week 2. Sutures out. The graft is now perfused by its own emerging vasculature. The clinical color may still look slightly different from the surrounding gingiva.
Week 3 to 4. Surface re-epithelialization is largely complete. The tissue tolerates gentle brushing with a soft toothbrush.
Month 1 to 3. Fibroblast remodeling is in full progress. Tissue tone and texture begin to mature.
Month 3 to 6. Final maturation. The grafted area now blends, biologically and clinically, with the surrounding gingiva.
Year 1 and beyond. Long-term stability data on autogenous CTG for root coverage is among the strongest in soft tissue surgery, with mean root coverage outcomes maintained over 10 to 20 year follow-ups (Zuhr et al., 2014).
For the procedural side of the harvest itself, including donor site management, suturing, and pain control, our 2015 piece on the four classic harvesting techniques is still the most practical entry point on Periospot.
Frequently asked questions
What is a connective tissue graft used for?
A connective tissue graft is used to cover exposed roots in cases of gingival recession, to increase the thickness of keratinized gingiva, and to augment soft tissue volume around teeth and dental implants. It is one of the most predictable procedures in periodontal plastic surgery.
How is a connective tissue graft harvested?
The graft is most often harvested from the lateral palate between the canine and the first molar, using a trap-door, single-incision, or Harris-style approach. The harvest preserves the surface epithelium at the donor site, which closes by primary intention and heals with less patient morbidity than a free gingival graft donor site.
How long does a connective tissue graft take to heal?
The visible clinical healing is largely complete by six to eight weeks. The full biological maturation of the graft, including fibroblast remodeling and integration into the surrounding gingiva, continues for three to six months.
What is the difference between a subepithelial CTG and a free gingival graft?
A subepithelial CTG is harvested from underneath the surface epithelium and contains only connective tissue. A free gingival graft is harvested as a single piece that includes both epithelium and connective tissue. The subepithelial CTG produces a better esthetic blend at the recipient site, and its donor site heals faster and with less patient discomfort.
Are there non-autogenous alternatives to a connective tissue graft?
Yes. Xenogeneic collagen matrices and acellular dermal matrices can replace an autogenous CTG in many indications, particularly for soft tissue thickening. Outcomes for root coverage remain slightly better with autogenous tissue, but non-autogenous materials eliminate the second surgical site and reduce patient morbidity.
Why does deepithelialization technique matter?
Because the choice of deepithelialization method determines how much epithelium actually leaves the graft. Cadaver evidence has shown that an intraoral bone scraper removes approximately 98% of the graft epithelium, a diamond bur about 88%, and an extraoral scalpel only about 59% (Bara-Gaseni et al., 2024). Residual epithelium can influence healing and the long-term cosmetic appearance of the graft.
Histologic storytelling

The future of dental education is teaching clinicians to see the biology underneath the procedure.
I never did go back to making sashimi at home. But the lesson stayed with me.
The knife was beautiful. The book was excellent. The videos were better than any course I had ever bought. None of it mattered until I understood what was actually happening between the steel and the fish. Not the technique. The biology of the cut. The grain. The temperature. The way the muscle fibers wanted to separate.
Connective tissue grafting works the same way.
The instruments matter, the courses matter, the protocols matter. They are the table stakes. They are the Yanagiba on the shelf and the book on the counter. What separates clinicians who get consistent root coverage from clinicians who do not is rarely the kit. It is whether you can see what the microscope sees. Whether you can predict, before the first incision, how that specific piece of tissue will integrate, revascularize, and mature. Whether you can read the biology underneath the procedure the same way a chef reads the grain of a piece of fish.
This is the thinking behind Periospot AI. Not a tool that replaces clinical judgment. A tool that makes the invisible biology visible at the moment of the decision, so the next generation of clinicians does not have to learn it the way most of us did, slowly, through cases that did not behave the way the book said they would.
The future of dental education is not only showing the procedures.
It is teaching clinicians to see the biology underneath.
Histologic storytelling. One millimeter at a time.
And next time you order sashimi, take a closer look at the cut. There is more biology on that plate than most courses ever teach.
References
Bara-Gaseni, N., Jorba-Garcia, A., Alberdi-Navarro, J., et al. (2024). Histological assessment of a novel de-epithelialization method for connective tissue grafts harvested from the palate. An experimental study in cadavers. Clinical Oral Investigations. https://doi.org/10.1007/s00784-024-05734-y
Maia, V. T. G., Kahn, S., de Souza, A. B., & Fernandes, G. V. O. (2021). Deepithelialized connective tissue graft and the remaining epithelial content after harvesting by the Harris technique: A histological and morphometrical case series. Clinical Advances in Periodontics, 11(3), 150–154. https://doi.org/10.1002/cap.10151
Tavelli, L., Barootchi, S., Greenwell, H., & Wang, H. L. (2019). Autogenous soft tissue grafting for periodontal and peri-implant plastic surgical reconstruction. Journal of Periodontology, 90(10), 1163–1180. https://doi.org/10.1002/JPER.19-0103
Zuhr, O., Bäumer, D., & Hürzeler, M. (2014). The addition of soft tissue replacement grafts in plastic periodontal and implant surgery: Critical elements in design and execution. Journal of Clinical Periodontology, 41(Suppl 15), S123–S142. https://doi.org/10.1111/jcpe.12185
Zuhr, O., Rebele, S. F., Schneider, D., Jung, R. E., & Hürzeler, M. B. (2014). Tunnel technique with connective tissue graft versus coronally advanced flap with enamel matrix derivative for root coverage. Journal of Clinical Periodontology, 41(6), 582–592.
Comments
0 totalLoading comments...
Related Articles

4 connective tissue harvesting techniques that may help you in your daily practice
16 min read
Root Coverage Procedures, My First Time! 8 Golden Rules on Esthetic Plastic Periodontal Surgery.
14 min read![Bundle Bone: The Complete Guide to Gap Management & Immediate Implants [2026 Update]](/_next/image?url=https%3A%2F%2Fajueupqlrodkhfgkegnx.supabase.co%2Fstorage%2Fv1%2Fobject%2Fpublic%2Fimages%2Fposts%2Fbundle-bone-the-complete-guide-to-gap-management-immediate-implants%2Fregenerated-1768916026108.jpeg&w=828&q=75)
